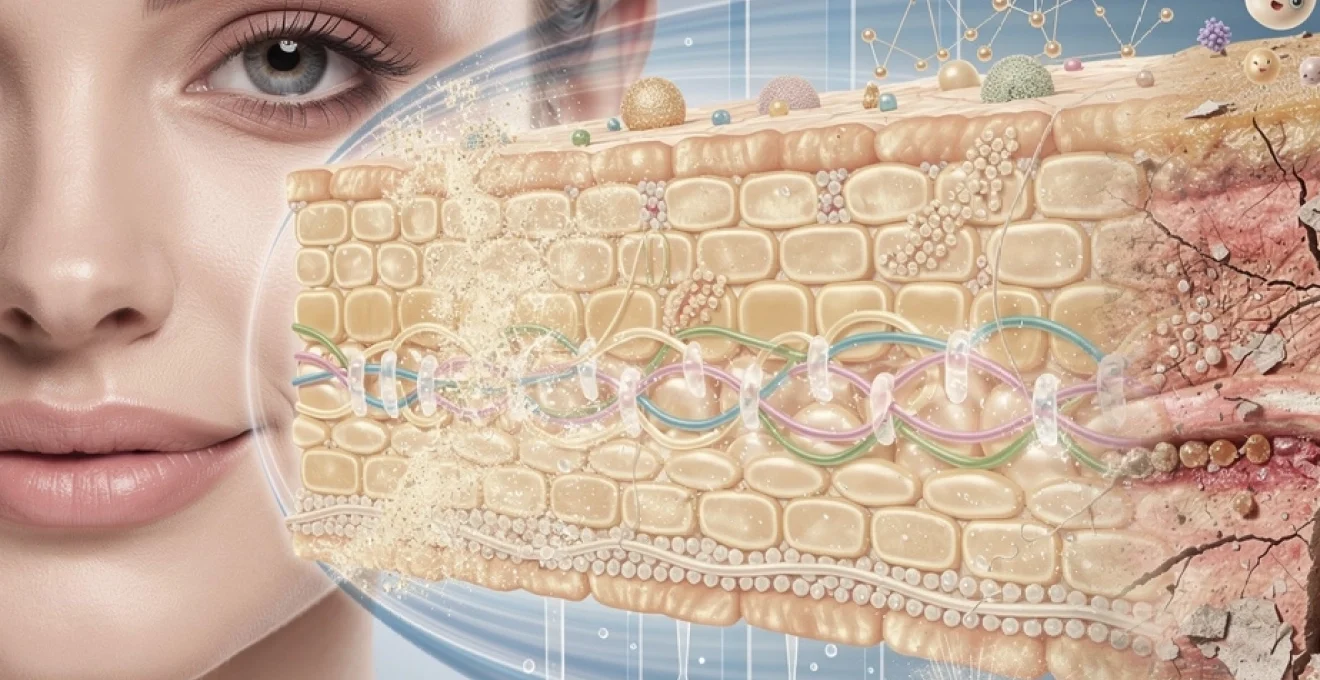
Your skin is far more than a simple covering for your body—it’s a sophisticated, multilayered organ that performs countless vital functions every second of every day. At the forefront of this biological marvel stands the skin barrier, a remarkably thin yet incredibly resilient structure that serves as your body’s first line of defence against environmental threats while simultaneously preventing precious moisture from escaping. Understanding the intricate mechanisms that govern this protective shield is essential for anyone seeking to maintain optimal skin health, whether you’re battling chronic conditions like eczema or simply aiming to preserve a healthy, radiant complexion. Recent research has revealed that this barrier is not merely a passive wall but an active, dynamic system involving complex biochemical processes, immune responses, and microbial interactions that work in concert to protect your wellbeing.
Stratum corneum architecture and lipid bilayer composition
The stratum corneum represents the outermost layer of the epidermis and serves as the primary structural component of the skin barrier. This remarkable layer, measuring only 10-20 micrometres in thickness—roughly one-fifth the width of a human hair—is responsible for the majority of the skin’s protective functions. The stratum corneum employs what scientists often describe as a “brick and mortar” architecture, where flattened, protein-rich skin cells called corneocytes act as the bricks, whilst a sophisticated matrix of lipids functions as the mortar holding everything together.
This architectural design isn’t merely coincidental; it represents millions of years of evolutionary refinement. The corneocytes themselves are essentially dead cells that have undergone a transformation process called cornification, during which they lose their nuclei and organelles whilst simultaneously filling with keratin proteins. These protein-packed cells are then embedded within the lipid matrix, creating a structure that is both flexible enough to accommodate movement and robust enough to withstand considerable environmental stress. Research conducted in 2023 indicates that a healthy stratum corneum contains approximately 15-20 layers of these corneocytes, though this number can vary depending on body location and individual factors.
Ceramides, cholesterol, and free fatty acids in the extracellular matrix
The lipid matrix that binds corneocytes together consists of three primary components: ceramides, cholesterol, and free fatty acids. These lipids must exist in a precise ratio—approximately 1:1:1—for optimal barrier function. Ceramides alone account for roughly 50% of the lipid content and are the most diverse group, with at least twelve distinct ceramide subclasses identified in human skin. These waxy lipids are particularly crucial because they form the foundational structure of the lipid lamellae, the organized sheets that create the barrier’s waterproofing properties.
Cholesterol, whilst often vilified in dietary contexts, plays an indispensable role in skin barrier function. It fills gaps between ceramides and helps maintain the fluidity and flexibility of the lipid layers. Without adequate cholesterol, the barrier becomes rigid and prone to cracking. Free fatty acids, meanwhile, contribute to the skin’s slightly acidic pH and provide additional structural support. Studies have demonstrated that even minor deviations from the optimal lipid ratio can result in measurable increases in trans-epidermal water loss, highlighting the precision required for proper barrier function.
Corneocyte envelope structure and filaggrin breakdown products
Each corneocyte is surrounded by a specialized structure called the cornified envelope, an exceptionally tough protein shell approximately 15 nanometres thick that provides mechanical strength to the cell. This envelope is composed primarily of involucrin, loricrin, and other structural proteins that have been cross-linked through enzymatic reactions to create an almost indestructible barrier. The cornified envelope is so resilient that it can withstand boiling in solutions containing strong detergents—a testament to its protective capabilities.
Filaggrin, a protein whose name derives from “filament aggregating protein,” plays a pivotal role in barrier function beyond its structural contributions. As corneocytes mature, filaggrin is progressively broken down into smaller amino acids and derivatives, collectively known as natural moisturizing factor (NMF). These breakdown products are hygroscopic, meaning
they attract and bind water within the stratum corneum, helping to keep the outer skin layers supple and resilient. Reduced filaggrin expression or accelerated filaggrin degradation leads to deficient NMF, impaired stratum corneum hydration, and increased susceptibility to irritation. This is one reason why filaggrin gene mutations are so strongly associated with atopic dermatitis and chronically dry, sensitive skin. When you notice your skin feeling tight, flaky, and easily reactive, it often reflects subtle changes in this microscopic protein–water balance.
Lamellar body secretion and lipid lamellae formation
While corneocytes provide the structural “bricks” of the skin barrier, lamellar bodies are responsible for delivering much of the “mortar.” These small, membrane-bound organelles are produced by keratinocytes in the upper spinous and granular layers of the epidermis. As cells migrate upward and approach the stratum corneum, lamellar bodies fuse with the plasma membrane and exocytose their contents into the extracellular space. These contents include precursor lipids, enzymes, and antimicrobial peptides that are essential for barrier formation.
Once secreted, the lipid precursors undergo enzymatic processing to form highly organized lamellar sheets composed primarily of ceramides, cholesterol, and free fatty acids. Think of these lamellae as tightly stacked layers of waterproof film interwoven between corneocytes, dramatically reducing permeability to water and irritants. Disruption in lamellar body formation or secretion—for example, due to genetic defects, chronic inflammation, or surfactant damage—results in incomplete or disorganized lipid lamellae. Clinically, this presents as increased transepidermal water loss, rough texture, and a higher risk of dermatitis. Supporting the skin barrier with topical lipid-replenishing products can, to an extent, compensate for impaired lamellar body function.
Transglutaminase activity in cornified envelope cross-linking
The transformation of a living keratinocyte into a tough, flattened corneocyte requires extensive protein cross-linking, a process largely driven by the enzyme family known as transglutaminases. These calcium-dependent enzymes catalyse the formation of covalent bonds between structural proteins such as involucrin, loricrin, and small proline-rich proteins, ultimately generating the insoluble cornified envelope. You can imagine transglutaminases as molecular riveters, fastening the structural framework of the cell into a rigid, protective shell.
Dysfunction in epidermal transglutaminases, whether genetic or acquired, leads to fragile, poorly cornified corneocytes and a compromised skin barrier. Certain rare disorders, like lamellar ichthyosis, are directly linked to transglutaminase mutations and illustrate how vital this cross-linking is: affected individuals exhibit severe scaling and markedly increased water loss. Even in less extreme scenarios, suboptimal enzyme activity can contribute to subtle barrier weakness and chronic dryness. Maintaining a healthy epidermal environment—adequate calcium gradients, controlled inflammation, and gentle skincare practices—helps preserve normal transglutaminase function and, by extension, robust cornified envelopes.
Trans-epidermal water loss and barrier permeability function
One of the most critical roles of the skin barrier is regulating the movement of water between the body and the environment, a process quantified as trans-epidermal water loss (TEWL). TEWL reflects the amount of water that passively diffuses from the deeper epidermal layers through the stratum corneum and evaporates from the skin surface. In healthy skin, TEWL is relatively low—typically around 5–15 g/m²/hour, depending on body site and climate—indicating that the barrier is effectively conserving internal hydration. When TEWL rises, it signals increased permeability and an impaired barrier, even if the skin still appears normal to the naked eye.
Why does this matter for overall skin health? Elevated TEWL is associated with dryness, irritation, and an increased risk of inflammatory skin conditions such as atopic dermatitis and irritant contact dermatitis. A leaky barrier also allows greater penetration of potential allergens and pollutants, which can trigger or exacerbate immune responses. Measuring TEWL has therefore become a valuable tool in both research and clinical practice for assessing barrier integrity and the efficacy of barrier-repair skincare routines. If your goal is long-term skin resilience, reducing excessive TEWL is just as important as adding moisture from the outside.
Aquaporin-3 expression and dermal-epidermal water transport
While lipids and corneocytes regulate water loss at the surface, specialised membrane channels known as aquaporins help manage water transport within the epidermis itself. Aquaporin-3 (AQP3), highly expressed in the basal and spinous layers of the epidermis, facilitates the movement of water and small solutes like glycerol between keratinocytes. You can think of AQP3 as a network of microscopic plumbing that helps distribute hydration from the dermis up toward the outer layers of the skin. This internal water flow is vital for maintaining cell viability, supporting enzyme activity, and enabling orderly differentiation.
Animal and human studies have shown that reduced AQP3 expression correlates with decreased skin hydration, delayed barrier recovery, and increased susceptibility to irritation. Conversely, enhancing AQP3 activity or glycerol availability can improve stratum corneum hydration and elasticity. Some moisturisers leverage this pathway by including humectants such as glycerin that both attract water and serve as substrates for AQP3-mediated transport. When you consistently use a well-formulated hydrating routine, you are not only adding surface moisture but also supporting these deeper water channels that underpin a healthy skin barrier.
Natural moisturising factor and stratum corneum hydration
Natural moisturising factor (NMF) is a collective term for a mixture of low-molecular-weight, water-soluble compounds derived largely from filaggrin breakdown. These include amino acids, pyrrolidone carboxylic acid (PCA), urea, lactate, and various inorganic ions. NMF resides primarily inside corneocytes, where it acts like an internal sponge, drawing and holding water to keep these “bricks” of the barrier plump and flexible. In a sense, the stratum corneum is like a well-packed bookshelf: NMF keeps each “book” (corneocyte) from becoming too dry and brittle, which would otherwise lead to cracking and microfissures.
When NMF levels drop—due to genetic filaggrin deficiency, excessive washing with harsh surfactants, low humidity, or ageing—the stratum corneum becomes dehydrated and rigid. Clinically, this is experienced as roughness, flaking, and sometimes a stinging sensation when products are applied. Supporting NMF can be approached in two ways: by preserving the skin’s own production (for example, limiting over-cleansing and maintaining an optimal pH) and by supplementing NMF components topically. Moisturisers that contain amino acids, PCA, urea, or lactates can help restore this internal water-binding capacity and are particularly beneficial for dry, barrier-compromised skin.
Occludin and claudin tight junction proteins in barrier integrity
Although the stratum corneum is the most visible component of the skin barrier, deeper cellular junctions also play a crucial role in controlling permeability. In the granular layer of the epidermis, tight junctions function as seals between adjacent keratinocytes, regulating the paracellular passage of water, ions, and solutes. These tight junctions are composed of transmembrane proteins such as occludin and members of the claudin family, anchored to a cytoplasmic scaffold of zonula occludens proteins. Together, they form a continuous belt-like structure around cells, comparable to the grout that seals tiles and prevents leaks.
Disruption of occludin or claudin expression—seen in conditions like atopic dermatitis and psoriasis—weakens this inner barrier layer, increasing both TEWL and susceptibility to irritants. Emerging research suggests that tight junction integrity is also influenced by external factors such as UV radiation, surfactants, and low humidity. From a practical standpoint, this underscores why a comprehensive barrier-support approach matters: it’s not only about the outer lipid layers, but also about preserving the health of deeper cell–cell connections. Using gentle cleansers, daily broad-spectrum sunscreen, and anti-inflammatory skincare ingredients helps protect these delicate protein complexes and maintain overall barrier function.
Antimicrobial peptides and immune defence mechanisms
The skin barrier is not just a physical shield; it also operates as an active immunological frontier equipped with potent antimicrobial and signalling systems. Keratinocytes, once thought to be passive structural cells, are now recognised as dynamic immune sentinels capable of producing antimicrobial peptides (AMPs), cytokines, and chemokines in response to environmental cues. This chemical defence layer works in tandem with the physical barrier to prevent colonisation by pathogenic microbes while supporting a balanced skin microbiome. When we talk about “skin resilience,” we’re also referring to this finely tuned interplay between structural integrity and immune vigilance.
Antimicrobial peptides, such as human beta-defensins and cathelicidin LL-37, act as natural antibiotics produced by your own body. They can directly kill bacteria, viruses, and fungi by disrupting their membranes, and they also modulate immune cell activity and inflammation. Importantly, AMP expression is tightly regulated; too little leaves the skin vulnerable to infection, while excessive or dysregulated production can contribute to chronic inflammation. Understanding these mechanisms helps explain why some individuals are more prone to recurrent infections or inflammatory flares when their skin barrier is compromised.
Human beta-defensin and cathelicidin LL-37 production
Human beta-defensins (hBDs) and cathelicidin LL-37 are among the best-characterised AMPs in the epidermis. These small, cationic peptides are produced by keratinocytes, sebocytes, and immune cells in response to stimuli such as microbial presence, mechanical injury, or UV exposure. Once secreted, they bind to negatively charged microbial membranes, creating pores that lead to rapid cell death—like tiny molecular spears breaching an enemy’s armour. Beyond their direct antimicrobial action, hBDs and LL-37 also recruit immune cells, promote wound healing, and influence angiogenesis.
In healthy skin, baseline levels of these peptides help contain potentially harmful microbes while allowing commensal organisms to coexist. However, in conditions like atopic dermatitis, LL-37 expression is often reduced, contributing to recurrent Staphylococcus aureus colonisation and infection. Conversely, in psoriasis and rosacea, AMP levels can be abnormally high and pro-inflammatory. From a skincare perspective, maintaining a stable, intact barrier indirectly supports balanced AMP production by reducing chronic irritation and the constant need for heightened immune activation.
Toll-like receptor signalling in keratinocyte immune response
How does the skin recognise when it is under microbial threat? One key mechanism involves toll-like receptors (TLRs), a family of pattern-recognition receptors expressed on keratinocytes, Langerhans cells, and other cutaneous immune cells. TLRs detect conserved molecular motifs on bacteria, viruses, and fungi—such as lipopolysaccharides or viral RNA—and trigger intracellular signalling cascades that lead to cytokine release and AMP production. In this way, keratinocytes behave much like innate immune cells, rapidly sensing danger and coordinating early defence responses.
Balanced TLR activation is crucial. Excessive or chronic stimulation, often due to a persistently disrupted skin barrier or dysbiotic microbiome, can drive ongoing inflammation and contribute to diseases like acne, rosacea, and atopic dermatitis. On the other hand, insufficient responsiveness may impair the ability to fend off pathogens. Gentle skincare that avoids harsh irritants and preserves barrier integrity helps maintain appropriate TLR signalling. Emerging therapies also target specific TLR pathways in an effort to modulate cutaneous immune responses more precisely in inflammatory skin disorders.
Skin microbiome balance and staphylococcus epidermidis colonisation
The skin barrier coexists with a rich ecosystem of microorganisms collectively known as the skin microbiome. Far from being mere contaminants, many of these microbes play beneficial roles in barrier function and immune regulation. A prime example is Staphylococcus epidermidis, a common commensal bacterium that colonises healthy skin. S. epidermidis can inhibit the growth of pathogenic species such as Staphylococcus aureus by producing bacteriocins and stimulating AMP production in keratinocytes. You might think of it as a resident security team that helps keep more dangerous intruders in check.
Disturbances in microbiome composition—driven by overuse of broad-spectrum antibiotics, harsh cleansers, or chronic inflammation—can reduce beneficial colonisation by species like S. epidermidis. This imbalance, or dysbiosis, is increasingly linked to conditions such as acne, atopic dermatitis, and seborrhoeic dermatitis. Strategies that support a diverse, stable microbiome, including pH-appropriate cleansers and barrier-friendly moisturisers, therefore contribute indirectly to stronger immune defence. Future skincare may increasingly incorporate prebiotics and postbiotics designed specifically to nurture protective microbes and restore microbial equilibrium.
Langerhans cell activation and antigen presentation pathways
Embedded within the epidermis are specialised dendritic cells known as Langerhans cells, which function as professional antigen-presenting cells. They extend long processes between keratinocytes, constantly sampling the microenvironment for foreign substances such as microbial components, allergens, and toxins. When Langerhans cells encounter a relevant antigen, they process it and migrate to regional lymph nodes, where they present it to T cells and initiate adaptive immune responses. This pathway bridges the innate and adaptive arms of immunity and is essential for mounting targeted, long-term defence.
However, Langerhans cell activation is a double-edged sword. In a healthy barrier, activation is proportionate and controlled. When the barrier is chronically disrupted—allowing repeated penetration of allergens or irritants—Langerhans cells may drive persistent T-cell activation, contributing to allergic contact dermatitis and chronic eczema. Maintaining an intact, well-hydrated stratum corneum reduces unnecessary antigen exposure and helps prevent this cycle of over-activation. This is one reason why consistent, barrier-supportive skincare can reduce sensitivity over time, even in individuals who are prone to reactive skin.
Ph gradient regulation and acid mantle maintenance
The outer surface of healthy skin is mildly acidic, typically with a pH between 4.0 and 5.5, forming what is commonly referred to as the acid mantle. This acidic environment is not incidental; it is fundamental to skin barrier integrity, microbiome balance, and enzyme activity. As you move deeper into the epidermis, pH gradually approaches neutral, creating a vertical pH gradient that orchestrates numerous biochemical processes. Enzymes involved in lipid processing, desquamation, and antimicrobial defence all have optimal activity ranges along this gradient.
Several factors contribute to acid mantle formation, including the breakdown of phospholipids into free fatty acids, the conversion of filaggrin into acidic NMF components, and the presence of lactic acid from sweat. A properly maintained acid mantle supports the organisation of lipid lamellae, stabilises tight junctions, and favours the growth of beneficial microbiota while inhibiting many pathogenic species. When this pH balance is disrupted—often by frequent washing with alkaline soaps, overuse of exfoliants, or certain dermatological conditions—barrier function and microbial homeostasis are quickly affected. You may notice this as increased dryness, stinging, or a tendency to break out.
From a practical standpoint, preserving the skin’s natural pH is one of the simplest and most effective ways to support long-term barrier health.
Choosing pH-balanced cleansers (usually in the 4.5–5.5 range), avoiding prolonged exposure to very hot water, and using leave-on formulations that respect or gently restore acidity can all help maintain the acid mantle. For individuals with sensitive or barrier-impaired skin, incorporating mildly acidic toners or serums can aid in re-establishing an optimal pH gradient. Ultimately, when you protect the skin’s acid mantle, you make every other aspect of your skincare routine more effective, from moisturisers to active treatments.
Barrier disruption pathology in atopic dermatitis and eczema
Atopic dermatitis (AD) and other eczematous conditions provide a vivid example of what happens when the skin barrier is chronically compromised. Rather than being purely inflammatory or purely structural diseases, these disorders reflect an intricate interplay between genetic predisposition, barrier dysfunction, immune dysregulation, and environmental triggers. Many individuals with AD carry filaggrin gene mutations, leading to reduced NMF, impaired cornified envelopes, and increased TEWL even in non-lesional skin. In practical terms, this means their skin is inherently more permeable and vulnerable to irritants, allergens, and microbes.
Once the barrier is weakened, exposure to environmental antigens more readily activates Langerhans cells and T cells, driving the characteristic Th2-skewed immune response seen in atopic dermatitis. Cytokines released during flares—such as IL-4 and IL-13—further downregulate filaggrin and lipid synthesis, creating a vicious cycle of barrier damage and inflammation. Clinically, this manifests as dry, intensely itchy patches, often with lichenification and secondary infection. Even between visible flares, subtle barrier abnormalities persist, which is why ongoing maintenance therapy is now a central concept in modern AD management.
Eczema outside the classical atopic pattern—such as irritant contact dermatitis, allergic contact dermatitis, or nummular eczema—also involves barrier disruption, albeit through somewhat different mechanisms. Repeated exposure to harsh detergents, solvents, or occlusive environments can strip lipids and damage corneocytes, leading to micro-cracks and increased TEWL. In allergic forms, specific haptens penetrate the impaired barrier, are recognised by Langerhans cells, and elicit a T-cell mediated response upon re-exposure. Regardless of subtype, the unifying theme is clear: without a competent skin barrier, environmental factors gain exaggerated access to the immune system, and inflammation becomes much easier to trigger.
Effective management of atopic dermatitis and eczema therefore targets both inflammation and the underlying barrier defect. While topical corticosteroids and calcineurin inhibitors address immune overactivity, regular use of barrier-repair emollients, avoidance of known irritants, and consistent pH-appropriate cleansing address the structural side of the disease. For many patients, adopting a daily, proactive moisturising regimen even when the skin appears “clear” significantly reduces flare frequency and improves quality of life. In other words, treating the barrier is not simply cosmetic; it is central to disease control.
Topical barrier repair strategies using emollients and occlusives
Given the complexity of the skin barrier, it might seem daunting to influence its function from the outside. Yet well-designed topical formulations can meaningfully support and restore barrier integrity. Two of the most important categories are emollients and occlusives, which work in complementary ways. Emollients, often composed of lipids and lipid-like substances (such as ceramides, squalane, and certain plant oils), fill in microscopic gaps between corneocytes, smoothing the surface and improving flexibility. Occlusives, including petrolatum, mineral oil, and some silicones, form a semi-occlusive film over the skin, significantly reducing TEWL by slowing water evaporation.
In barrier-compromised skin, combining these strategies is often most effective. An emollient rich in physiological lipids—ceramides, cholesterol, and free fatty acids in ratios that mimic the natural barrier—can help rebuild the “mortar” of the stratum corneum. Layering or formulating this with an occlusive agent creates a protective seal, allowing time for the skin to restore its own lipid production and cellular architecture. Clinical studies have shown that petrolatum-based occlusives can reduce TEWL by up to 99%, while ceramide-containing moisturisers improve barrier recovery after experimental damage and reduce symptoms in conditions like atopic dermatitis.
How can you translate this science into a daily routine? For most skin types, a gentle, pH-balanced cleanser followed by a moisturiser that combines humectants (such as glycerin or hyaluronic acid), emollients (like squalane, shea butter, or ceramides), and light occlusives provides a solid foundation. In colder, drier climates or during barrier repair after irritation, you may benefit from a slightly richer, more occlusive formulation at night to lock in hydration. Applying moisturiser to slightly damp skin after bathing maximises water retention and supports the skin’s natural repair processes during sleep.
Equally important is avoiding unnecessary damage in the first place. Limiting over-exfoliation, steering clear of high-alcohol or strongly fragranced products if you are sensitive, and using broad-spectrum sunscreen daily all reduce the cumulative stress on your skin barrier. When active ingredients like retinoids or hydroxy acids are part of your routine, introducing them gradually and pairing them with robust barrier support can help you reap their benefits without tipping the balance toward inflammation. Ultimately, a skin barrier–centric approach to skincare recognises that virtually every product choice you make either supports or challenges this delicate, vital system—and that long-term skin health depends on keeping it strong.